EmeraMed: Targeting the cause of diseases.
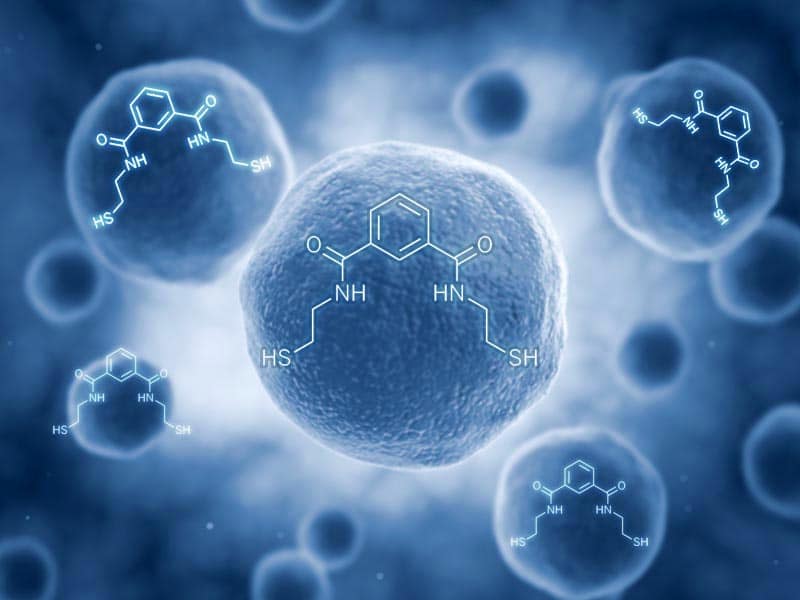
Oxidative stress releases free radicals. Emeramide captures free radicals, which are unstable molecules that can damage the cells. Additionally Emeramide captures the heavy metals mercury and iron which would otherwise create free radicals. Emeramide thereby helps restore normal function to the mitochondria and overall health.
COPD: A Phase 2a clinical trial has been completed in summer of 2018 and found Emeramide to be safe with no adverse effects.
Other conditions: Preclinical studies are ongoing for several other indications.
Covid-19: Covid-19 may have an association with oxidative stress and high iron levels, studies are underway to support our belief that Emeramide contributes to the treatment of Covid-19.